Research Article 
 Creative Commons, CC-BY
Creative Commons, CC-BY
Acute Allopregnanolone i.c.v. Modifies Aggressive Behavior Induced by pCPA Serotoninergic Depletion: Interplay with Serotonergic Activity, TPH2, GABAA α1, and GABAAδ mRNA Expression in the Olfactory Bulb
*Corresponding author: Ricardo Cabrera, INBIOMED, University of Mendoza, Huarpes 630, 5500 Mendoza-Argentina.
Received: December 13, 2024; Published: December 17, 2024
DOI: 10.34297/AJBSR.2024.25.003301
Abstract
Previously, we found that after a single i.p. administration of pCPA in male rats, aggressive behavior parameters were increased, serotonergic activity was reduced, and TPH2 and GABAAα1 expression was increased in the olfactory bulb. Here, we investigate the interaction of allopregnanolone with serotonergic activity, TPH2, GABAAα1, and GABAAδ mRNA expression in the olfactory bulb. To study allopregnanolone, a stereotactic surgical protocol was developed in which a fixed cannula was implanted in the third ventricle before i.p. administration of pCPA. This protocol assessed animals’ aggressive behavior in an intruder test six days after pCPA administration, and allopregnanolone (6μM) was administered i.c.v. 30 min before the test. Subsequently, the concentrations of 5-HT and 5-HIAA were determined in the olfactory bulb. Because allopregnanolone modulates the GABAergic system and the olfactory bulb has abundant GABAergic innervation, mRNA expression of GABAAα1 and GABAAδ subunits was determined in parallel with TPH2. We recovered aggressive behavioral parameters in pCPA-depleted animals and increased 5-HT, 5-HIAA, and serotonergic catabolism. In addition, GABAAα1 mRNA expression was decreased, whereas GABAAδ mRNA expression did not change in the olfactory bulb. These results suggest that allopregnanolone plays a role in aggressive behavior and module serotonergic and GABAergic in the olfactory bulb.
Keywords: Allopregnanolone, pCPA, Aggressive behavior, 5-HT, TPH2, GABAAα1, GABAAδ; Olfactory Bulb
Introduction
Serotonin (5- HT) plays a vital role in aggressive territorial behavior [3,6,44-46]. 5-The enzyme Tryptophan Hydroxylase (TPH) synthesizes HT in the raphe nuclei. The expressed TPH2 isoform has been described mainly in neurons of the raphe complex [36,49]. 5-HT-producing neurons in the raphe project their axons to the cortex, amygdala, hippocampus, basal ganglia, thalamus, hypothalamus, and Olfactory Bulb (OB) [Lesch & Waider, 2012; 33]. OB is an important serotonergic innervated structure, and we have recently demonstrated mRNA expression of the TPH2 isoform in OB [14]. GABAA receptors are found in principal neurons and local interneurons in the OB [34]. Mitral and tufted cells receive the primary GABA- ergic input from two populations of interneurons, periglomerular cells, and granule cells, respectively, via specific dendrodendritic synapses in the glomerular layer and the external plexiform layer [40]. At the same time, the sense of smell regulates social behaviors such as aggressive encounters in rodents [14,41].
The GABAA receptor is a Cl ion channel distributed throughout the brain [28,50]. There are seven leading families of receptor subunits (α, β, γ, δ, ε, θ, π) and some families with multiple isoforms (α1-6, β1-3, γ1-3). The neurophysiological and pharmacological properties of GABAA receptors depend on the subunit [28,38,50]. Combining these subunits and isoforms results in a functional division of GABAA receptors: synaptic (phasic) and extra synaptic (tonic). While synaptic receptors have an α1;2;3;6, β, γ, extra synaptic ones have an α4;5, β, δ [24,38]. However, the most abundant GABAA receptor-α subunits are 1 and 3, expressed in mitral and tufted cells in the OB, and have odor-discriminatory activities [34]. Furthermore, four weeks of isolation in male mice decreased the expression of the α1-isoform of the GABAA receptor compared to group-housed males [26]. Furthermore, the most abundant subunit of the GABAA receptor in adult neurons is the α1-subunit, which is also associated with rapidly declining cortical neuronal currents [4].
Allopregnanolone (Allo) (3α-hydroxy-5α-pregnan-20-one) is the primary active metabolite of progesterone. It is one of the best-studied neuroactive steroids and influences a wide range of behaviors: sexual, affective, cognitive, and aggressive. Aggressive behavior is influenced by various neurobiological factors such as neurotransmission systems and neuroactive steroids [15,23]. Neuro steroids act in two ways: fast action via ion channel receptors and slow action via cytosolic receptors that activate conventional genomic expression [10]. The reported effects of Allo are associated with the GABAA receptor, where Allo acts as a positive allosteric modulator [10]. Allo has a particular affinity for GABAA receptors with the δ-subunit present on extra synaptic GABAA receptors [5,7,8,29,43]. Several studies [38,42] show that exposure to neuroactive steroids, such as Allo, results in altered GABAA receptor structure, in some cases associated with changes in GABAA expression subunits [7], and these could have behavioral implications. Increased extra synaptic GABAA receptor expression increases sensitivity to neuro steroids by increasing tonic current [7,51]. In contrast, deficient expression of the extra synaptic GABAA receptor decreases sensitivity to neuro steroids or neuroactive steroids [38, 42]. Carver CM, et al., propose the hippocampus as the nucleus for mRNA expression of the δ-subunit in the brain. This expression has several functions for memory and learning. In our laboratory, we have demonstrated the modulatory effect of Allo in the hippocampus [13,14].
Another brain structure that resembles the plasticity of the hippocampus in the adult brain is the OB. In 2014, Cook, et al., demonstrated that the regulatory enzymes of neuro steroidogenesis are present in the hippocampus, cortex, and OB neurons. They also performed tasks related to memory, learning, and social interactions. However, there are no controlled studies linking the effects of neuroactive steroids in OB. Furthermore, increased aggressive behavior and decreased Allo levels have been found in mice living in social isolation [26]. While high, administered Allo concentrations reduce aggressiveness [31]. In moderately administered concentrations, Allo increases aggression [16]. This evidence prompted us to investigate the possible involvement of Allo in a model of aggressive behavior in rats induced by serotonergic i.p. depletion. To investigate this, we analyzed the parameters of aggressive behavior, serotonergic activity, TPH2, GABAAα, and GABAAδ mRNA expression in OB after acute i.c.v. Administration of Allo.
Methods and Materials
Subjects and Housing
Animals were housed in a temperature-controlled animal room (22+/-2℃) with a 12-h light-dark cycle (light from 07:00 to 19:00h) and artificial light (60-70 lumens). Food and water were provided ad libitum. Residents: Male Sprague-Dawley rats of 60 days and 350g (average weight). Intruders: Male Sprague-Dawley rats are 50 days old and weigh 280g (average weight). After the surgical procedure, all resident subjects were housed in boxes for six consecutive days to recover. We divided the subjects into four groups. Two received i.p. vehicle (control veh; control allo), and two received i.p. pCPA (pCPA veh; pCPA Allo). Intruder subjects remained in groups of 3 until the resident intruder test (RVI), after which they were euthanized. All animal experiments were approved by the Comité Institucional para el Cuidado y Uso de Ani-males de Laboratorio de la Universidad Nacional de Cuyo (CICUAL-UNCuyo) (Aval 82/2016), Mendoza, Argentina.
Reagents
The drugs used were: pCPA-methyl ester hydrochloride, allopregnanolone (3α-hydroxy-5α-pregnan-20-one) (SIGMA, St. Louis, MO, USA); penicillin G benzathine (Richet, Argentina); ketamine HCL (Holliday-Scott S.A, Argentina) and xylazine (Koening Laboratories, Argentina). pCPA methyl ester hydrochloride was dissolved in saline at pH 6.5 at a concentration of 300mg/kg [19]. Allo was first dissolved in propylene glycol at a concentration of 600μM. The dose of Allo used in the experiments, six μM, was obtained by serial dilution in sterile saline with 1% propylene glycol to the indicated concentration [14]. The Allo concentration was chosen following previous findings from our laboratory in a pharmacological range [13,14]. In addition, similar concentrations of Allo (5μM) have been reported for its inhibitory effect on GABAergic neuronal activity in the dorsal raphe nucleus through activation of the GABAA receptor [20]. Benzathine penicillin G(im) was resolved at a concentration of 1,200,000 IU (0.6μg; 72mg/rat) [1,10]. The reagents used in each procedure are described in the following sections.
Surgical Procedure
Rats were anesthetized with ketamine HCL (80mg/kg) and xylazine (4mg/kg). Confirmation of the level of anesthesia was established by loss of the postural, palpebral-palpebral, corner-palpebral, and foot reflexes and by the absence of tail pinch response. They were then placed in a stereotaxic position (David Kopf, USA) for surgical intervention. To avoid hypothermia, the animals were covered with a blanket during the surgical procedure; during recovery from anesthesia, the animal was covered with the same blanket. A cotton pad was placed over the animal’s eyes to avoid eye injury during the procedure. The implants were placed in the region of the third cerebral ventricle according to the atlas coordinates of Paxinos and Watson: AP 0.0mm (Bregma), L: 0mm, DV 9mm, dental ridge inclination: +5°. The cannulas were inserted 1 mm above the structure to avoid injuring the tissue. Stainless steel guide cannulas (13.5mmx0.80mmx38mm) were used, which were fixed to the skull with dental cement and a stainless-steel screw. The cannulas were closed with a stainless-steel wire at the end of the process to prevent them from becoming blocked. To avoid possible infections, all animals received a subcutaneous injection of benzathine penicillin G (i.m) of 1,200,000IU [1,10]. Subsequently, each animal was placed in a box with a bed of pine shavings on a pad of tissue paper, with food and water available until recovery of the anesthetic level was verified (we waited until the animal showed coordinated voluntary movements).
Drugs Administration
Animals were injected i.p. with pCPA 300mg/kg (pCPA veh; pCPA Allo) or saline (control veh or control Allo) and returned to their home cage for six days. On the day of the behavioral test, the experimental animals received Allo or veh i.c.v.(1μl) (Cáceres et al., 2020) into the third ventricle 30 min beforehand. Control animals were injected with sterile saline containing 1% propylene glycol [13] (Nishida, et al., 2020)-all i.c.v. Administrations were performed with a needle (0.5mm diameter) connected to a 10μl syringe (Hamilton et al.) and were performed gently (0.25μl/min) [1]. The needle was left in place for 60 s to allow diffusion of the drugs in the ventricular system; then, the experimenter removed it [14]. A schematic representation of the experimental procedure can be seen in Figure 1.
Behavioural Tests
The RVI test was used to examine aggressive behavior. The RVI test was conducted following Koolhaas et al. (2013). In summer, the RVI test involves the interaction of animals: the resident, who is the test animal, and the intruder, who is the interaction animal. An RVI test was conducted 6 days after the administration of pCPA between 15:00 and 18:00 (Corthell, 2013). The test was conducted in a wooden box containing wood crisps from the resident’s home cage. After each test, the box was cleaned with ethanol (10%) [9]. The Everio G-series GZ-MG330 from JVC was used to record all tests. The total time of the RVI test was 900 seconds. It was divided into two phases: the adaptation phase and the interaction phase. The first 300 seconds were considered the resident’s adaptation phase to the environment. The remaining 600 seconds were considered the interaction phase. At the beginning of the interaction phase, an intruder was placed on the opposite side of the resident at the end of the adaptation phase, and the interaction behavior was measured. To exclude a possible effect of pCPA on the locomotor activity of the animals, the distance traveled during the adaptation phase was evaluated. In the interaction phase, we assessed four main groups of behaviors: 1-non-social activity, i.e., the time of grooming, sniffing, and sitting; 2-social activity, i.e., the time of hetero grooming and hetero sniffing; 3-aggressive behavior, i.e., as a time of the chase, moving towards, upright posture and false mount, bite, clinch, clinch attack, lateral threat and keep down during the interaction phase; and 4-aggressive latency, i.e., the time before the first aggressive behavior.
5-HT and 5-HIAA Content Determination by HPLC in the Olfactory Bulb.
Tissue homogenization was performed according to [11]. Briefly, the tissue was collected in 400μl of 0.2N perchloric acid and then homogenized in a glass-glass homogenizer. The homogenate was centrifuged at 12000×g for 15min at four ℃ (Hermle, et al., model Z233MK-2), and the supernatant was injected into a High-Performance Liquid Chromatograph (HPLC) instrument coupled to electrochemical detection to measure 5-HT, 5-HIAA. Using bovine serum albumin as standard, the pellet was resuspended in 1N NaOH for protein quantification by the Bio-Rad Protein Assay (Bio-Rad Laboratories, Inc., Richmond, CA, USA). The contents of 5-HT and 5-HIAA were expressed as picograms per milligram of total protein. Ten microliters of each supernatant were injected into the HPLC system with the following setting: An isocratic pump (model PU-2080 Plus, Jasco Co. Ltd., Tokyo, Japan), a UniJet microbore column (MF-8912, et al.,) and an ampere metric detector (set at 650mV, 0.5nA; model LC-4C, BAS, West Lafayette, IN, USA). The mobile phase, containing 0.05 M NaH2PO4, 1.0mM 1-octanesulfonic acid, 0.27mM EDTA, 1.0%(v/v) tetrahydrofuran, and 4.0%(v/v) acetonitrile (CH3CN) (pH adjusted to 2.6) was pumped at a flow rate of 100μl/min. The level of neurotransmitters and metabolites was assessed by com paring the sample’s respective peak area and elution time with a reference standard, and the quantification was performed using a calibration curve for each neurotransmitter (Program et al., Tokyo, Japan). Under these experimental conditions, retention times were 33.3 for 5-HT and 25.6 for 5-HIAA. Standards, EDTA, and 1-octane sulfonic acid were purchased from Sigma-Aldrich, Inc. (St, et al.), and all other reagents were of analytical grade. 5-HIAA/5-HT turnover was expressed as a percentual ratio.
RNA Extraction and RT-PCR Analysis
According to the manufacturer’s specifications, total RNA from OB was extracted using the TRIzol™ reagent (Invitrogen-Life et al., Argentina). Integrity of the mRNA of the samples was confirmed by 1% agarose gel electrophoresis and staining with Syber Gold™ (Invitrogen-Life, et al., Argentina.) 10μg of total RNA was reverse transcribed at 37℃ using random hexamer primers and Moloney murine leukemia virus retro transcriptase (Invitrogen-Life et al., Argentina) in a 20μL reaction mixture. RNA was first denatured at 70℃ for 5 min in the presence of 2.5μg of random hexamer primers (Invitrogen). For the subsequent RT reaction, the following mixture was added: RT buffer [50mM Tris–HCl (pH 8.4), 75mM KCl, three mM MgCl2], 0.5mM dNTPs, five mM DTT, 200 units M-MLV Reverse Transcriptase (Invitrogen). The reaction was incubated at 37℃ for 50min.; next, the reaction was inactivated by heating at 70℃ for 15min. The cDNA was stored at-20℃. TPH2, GABAA α1, and GABAAδ mRNA levels were estimated by RT-PCR with a Corbett Rotor-Gene 6000 Real-Time Thermocycler (Corbett, et al., Australia) using rat-specific primers and reaction conditions described in Table 1. PCR reactions were performed using a Corbett Rotor-Gene 6000 Real-Time Thermocycler using Eva-GreenTM (Biotium, et al.) in a final volume of 20μL. The reaction mixture consisted of 2μL of 10×PCR Buffer, 1μL of 50mM MgCl2, 0.4μL of 10mM dNTP Mix (Invitrogen), 1μL of 20×Eva Green, 0.25μL of 5U/μL Taq DNA Polymerase (Invitrogen) 0.1μL of each 2.5mM primer (forward and reverse primers) and 10μL of diluted cDNA.
PCR reactions were performed at the conditions described in Table 1. Melt curve analysis was used to determine whether a specific amplified product was generated. Absolute time quantification was monitored by measuring the increase in fluorescence caused by the binding of Eva Green dye to double-strand DNA at the end of each amplification cycle. Relative expression was determined using the comparative quantitative visitation method of normalized samples about the expression of a calibrator sample, according to the manufacturer’s protocol [35]. Each PCR run included a no-template control and a sample without reverse transcriptase. All measurements were performed in duplicate. Reaction conditions and quantities of cDNA added were calibrated such that the assay response was linear concerning the amount of input cDNA for each pair of primers. RNA samples were assayed for DNA contamination by performing the different PCR reactions without prior reverse transcription. Relative levels of mRNA were normalized to the S16 reference gene. RT-PCR products were analyzed on 2% agarose gels containing 0.5mg/mL ethidium bromide, and a unique band of the approximately correct molecular weight corresponded with a unique peak in melt curve analysis. RT-PCR reactions were carried out for 40 cycles, with an initial step of 5min at 95 ºC followed by a three-step scheme: 30s at 95 ºC, 30s at the annealing temperature shown above for each primer pair, and a final step at 72ºC for 30s. Primers were designed with Beacon Designer 7.9 software.
Statistical Analysis
Comparisons between the means of the different groups were made using the one-way test ANOVA and the Tuckey post-hoc test. The significance level was set at p<0.05 for all statistical tests. Data were expressed as means ±S.E.M. Normality tests (Shapiro-Wilk) were performed for all variables studied; results showed deviations from normality were insignificant. All data were analyzed using the software application GraphPad Prism 9.0 (GraphPad et al., USA).
Results
Behavioral Tests
There was a significant reduction in distance travel (F(1,41)=31.02;p<0.0001) in the control group (non-pCPA) after acute Allo administration. Interestingly, Allo did not affect the distance traveled in the pCPA groups (Figure 2). Acute Allo administration significantly increased non-social activity (F(1,41)=3.758 p<0.01; p<0.001; p<0.0001) compared to all groups (Figure 3). In contrast, acute Allo administration resulted in a significant decrease in social activity compared to all groups (F(1,41)=30.65; p<0.05; p<0.01; p<0.001; p<0.0001) (Figure 4). Interestingly, acute Allo administration did not affect non-social and social activity in the pCPA group.
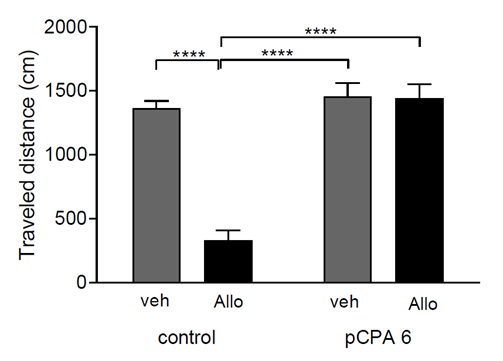
Figure 2: Effect of Allo on Locomotor Activity. Results are expressed as mean ±SEM of distance traveled in control veh (n=10); control allo (n=11); pCPA veh (n=9) and pCPA Allo (n=15). ****p<0.0001 for one-way ANOVA.
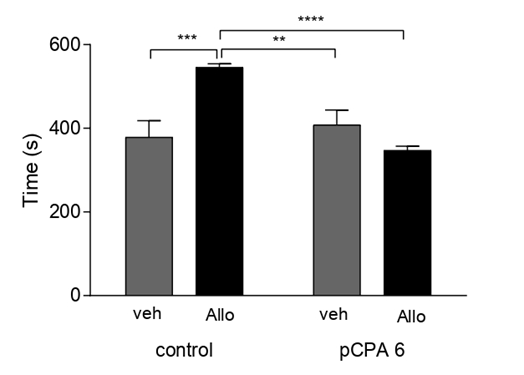
Figure 3: Effect of Allo on Nonsocial Behavior: Results are expressed as mean ±SEM of nonsocial time in second in control veh (n=10); control allo (n=11); pCPA veh (n=9) and pCPA Allo (n=15). **p<0.01; ***p<0.001; ****p<0.0001 for one way ANOVA.
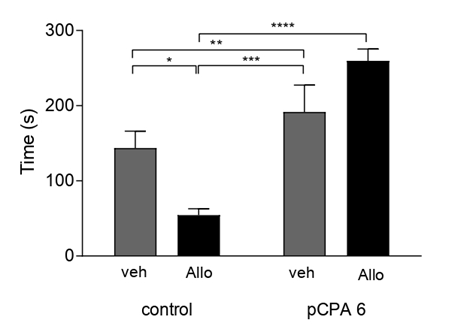
Figure 4: Effect of Allo on Social Behavior: Results are expressed as mean ±SEM of social time in second in control veh (n=10); control allo (n=11); pCPA veh (n=9) and pCPA Allo (n=15). *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001 for one way ANOVA.
It was found that acute Allo administration affected aggressive latency and aggressive behavior in the previously administered pCPA group. First, Allo had no effect on aggressive latency in the control groups. However, Allo significantly (p<0.01) increased aggressive latency in the pPCA groups. We also observed a significant increase in aggressive latency (p<0.0001) between the Allo-treated groups not treated with pCPA and those previously treated with pCPA after Allo administration. (All comparisons F (1,41)=83.97) (Figure 5). Second, we observed a significant decrease in aggressive behavior (F(1,41)=6.850; p<0.01) in the pCPA group after acute Allo administration (Figure 6).
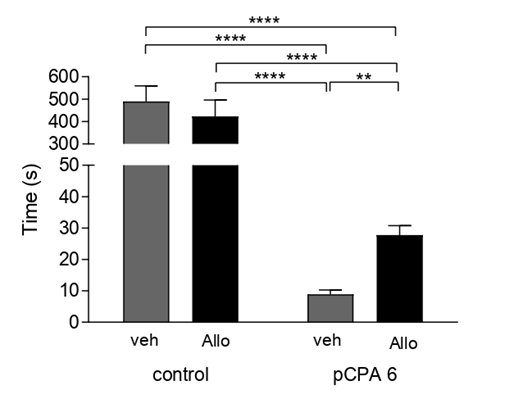
Figure 5: Effect of Allo on Aggressive Latency: Results are expressed as mean ±SEM of latency time in second in control veh (n=10); control allo (n=11); pCPA veh (n=9) and pCPA Allo (n=15). **p<0.01; ****p<0.0001 for one way ANOVA.
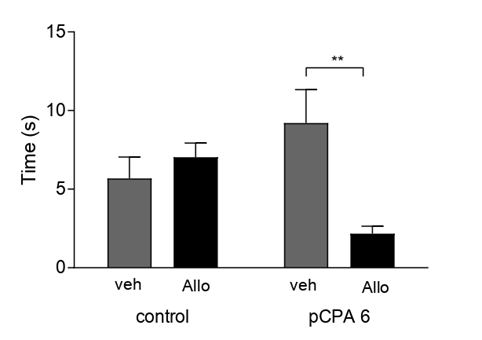
Figure 6: Effect of Allo in Aggressive Behavior: Results are expressed as mean ±SEM of aggressive time in second in control veh (n=10); control allo (n=11); pCPA veh (n=9) and pCPA Allo (n=15). **p<0.01 for one-way ANOVA.
5- HT and 5-HIAA Content Determination by HPLC in the Olfactory Bulb.
It was found that acute administration of Allo (Figure 7) induced a significant increase in the concentration of 5-HT (p<0.05) in OB, whether non-pCPA was previously administered. Similarly, we observed a significant increase (p<0.01) between the pCPA group and the Allo-non-pCPA group (all comparisons F (1,27)=18.49). The concentration of 5-HIAA metabolites increased significantly (F(1,27)=8.63; p<0.01) after the administration of Allo in the pCPA- treated groups, OB (Figure 8). In addition, we observed that acute Allo administration significantly increased (F(1,27)=10.47; p<0.01; p<0. 001) serotonergic activity (Figure 9).

Figure 7: Effect of Allo in 5-HT content determination by HPLC in the olfactory bulb: Results are expressed as mean ±SEM of pg/mg proteins in control veh (n=8); control allo (n=8); pCPA veh (n=7) and pCPA Allo (n=8). *p<0.05; **p<0.01 for one way ANOVA.
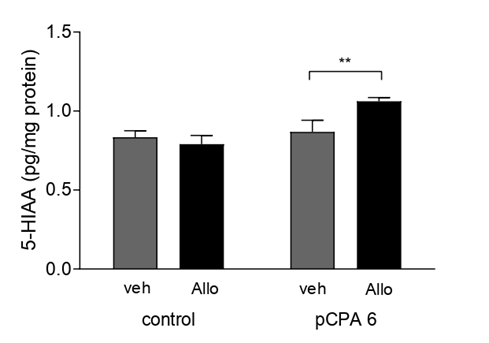
Figure 8: Effect of Allo in 5-HIAA content determination by HPLC in the olfactory bulb: Results are expressed as mean ±SEM of pg/mg proteins in control veh (n=8); control allo (n=8); pCPA veh (n=7) and pCPA Allo (n=8). **p<0.01 for one-way ANOVA.

Figure 9: Effect of Allo in Turnover 5-HIAA/5HT: Results are expressed as mean±SEM of %pg/mg proteins for control veh (n=8); control allo (n=8); pCPA veh (n=7) and pCPA Allo (n=8). **p<0.01; ***p<0.001 for one-way ANOVA.
RT-PCR Analysis
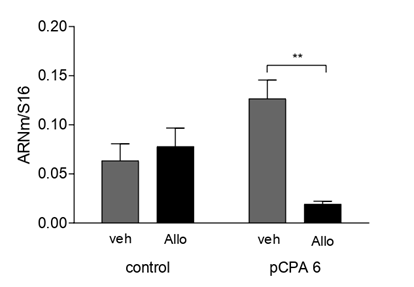
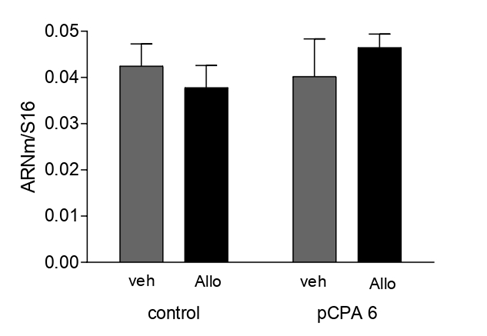
It was found that acute administration of Allo induced a significant decrease (F(1,27)=3.95; p<0.05) in TPH2 mRNA expression in OB (Figure 10). Similarly, Allo induced a significant decrease (F(1,27)=5.61; p<0.01) in GABAAα1 mRNA expression in OB (Figure 11). Interestingly, Allo did not affect GABAAδ mRNA expression (F(1,27)=0.0138)(Figure 12).

Figure 10: Effect of Allo in TPH2: Results are expressed as mean ±SEM of the relative expression units of rat TPH2 in control veh (n=8); control allo (n=8); pCPA veh (n=7) and pCPA Allo (n=8). *p<0.05 for one-way ANOVA.
Discussion
Social interaction, including aggressive behavior, is controlled by several neurobiological factors. Neuroactive steroids exert modulatory functions on neurotransmission systems involved in aggressiveness. In this work, we investigate the acute effects of Allo i.c.v. in a model of aggressive behavior in rats produced by serotonergic i.p. depletion. We investigated whether these effects occur in OB serotonergic activity, the structure with the most outstanding value for social interaction in rodents and whether they correspond to membrane responses or changes in the expression of specific genes. We examined Allo concentrations previously reported [14],[20] and demonstrated that Allo altered aggressive behavior parameters in animals with serotonergic depletion by pCPA. Specifically, Allo increased aggressive latency and decreased aggressive behavior. These results are consistent with the qualitatively less aggressive behaviors that Takahashi and Miczek (2014) suggested, as the animals showed a marked tendency toward social integration. Other reports [16] describe an increase in aggression after i.p. administration of Allo. However, it is important to note that biphasic effects of neuroactive steroids are commonly observed [31]. One possible explanation is the concentration of Allo and the administration via. We also observe that in subjects without serotonergic depletion, an increase in aggressive latency and a decrease in aggressive behavior are associated with increased sedation. Consistent with the suggestion of sedation in animals without depletion, the parameters of locomotor activity and nonsocial and social interaction did not appear to be differences between groups with serotonergic depletion mediated by pCPA, suggesting a decrease in aggression exclusively mediated by Allo in these groups.
Allo increased the concentrations of 5-HT and 5-HIAA in OB in subjects with serotonergic fatigue. In addition, we observed an increase in the turnover rate of Allo action in serotonergic system catabolism. Our results are consistent with other studies [39] showing that i.c.v. Administration of Allo in female rats increases the firing rate of raphe nucleus serotonergic neurons in vivo. They suggest a direct effect of Allo on serotonergic release in neurons of different brain regions. One possible explanation is that Allo mediates its effect through allosteric modulation of GABAA receptors in raphe [18]. Although GABAA receptors are distributed throughout the brain, it has also been documented that modulation of firing from the raphe nucleus to OB is the product of a delicate balance between inhibitory 5-HT1A/1B autoreceptors (McDevitt, 2011) and GABAA receptors in neurons of the raphe nucleus [18]. Previously, we showed that TPH2 expression increased in OB after pCPA [14]. Interestingly, Allo decreased TPH2 expression in OB. Immunohistochemical studies are needed to confirm the presence of 5-HT-producing neurons in the Olfactory Bulb (OB). However, our findings are consistent with previous research [2,21,25,32,37,53], which demonstrated TPH2 mRNA expression in the cortex, thalamus, hypothalamus, hippocampus, amygdala, and cerebellum in both humans and other species. Immunohistochemical studies are needed to confirm the presence of 5-HT-producing neurons in the Olfactory Bulb (OB). However, our findings are consistent with previous research [2,21,25,32,37,53], which demonstrated TPH2 mRNA expression in the cortex, thalamus, hypothalamus, hippocampus, amygdala, and cerebellum in both humans and other species.
GABAA receptors play a role in olfactory [48], and the changes in aggressive behavior observed with neuroactive steroid treatment may be explained by the diversity of GABAA receptor subunits and conformations [26,16,26]. Moreover, circulating concentrations of neuroactive steroids such as Allo may alter GABAAδ receptor subunit expression [51,24], and GABAAα1 is differentially expressed in mitral cells of OB [34]. Specifically, we investigate whether Allo modifies the differential expression of GABAAα1 mRNA in synaptic receptors and GABAAδ in extrasynaptic receptors after acute administration. We observed that GABAAα1 mRNA expression was altered in the presence of Allo, but GABAAδ expression in OB was not. Interestingly, GABAAα1 expression was altered by Allo because [34] reported that OB cells preferentially express GABAAα1 over other α-subunits.
Others [7,51] found that GABAAδ-mRNA expression was modified after exposure to neuroactive steroids in other brain regions, but prolonged exposure was required (Pinna et al., 2004). A comprehensive analysis of these results would suggest that Allo i.c.v. Reduces GABAAα1 subunit mRNA levels in OB, offsetting the high requirement for synthesis of this subunit caused by serotonergic depletion in an animal with aggressive behavior. Allo, in the OB, through positive GABAergic modulation, could induce a more intense pattern of neuronal activity, which exerts a facilities comparison between the stored memories and the new ones acquired in the test, may lead to more straightforward discrimination of the opponent and changes in behavior associated with the pattern of adaptive aggressive behavior of the species. Therefore, in rodents where odor is the first sensory information to establish social interaction, Allo may contribute to the mechanisms of olfactory discrimination mediated by GABAergic and serotonergic systems in OB. The tendency not to attack and interact evenly with intruders must have some relationship with GABAA receptor subunits in OB after Allo administration, which would elicit differential olfactory discrimination of chemical signals in the context of territory.
Conclusion
Thus, we conclude that acute administration of Allo in a serotonergic system depleted by pCPA leads to decreased aggressive behavior without affecting locomotion, nonsocial, and social activity. We have demonstrated decreased TPH2 mRNA expression in OB and shown that GABAAα1 subunit mRNA is decreased in OB after Allo administration. Our data provide evidence for the modulatory neuroactive role of steroids in behavior and demonstrate that OB is a relevant structure for understanding the neurobiological complexity of aggressive behavior.
Funding
The work was supported by grants from the National Research Council of Argentina CONICET (PIP.2022-2024) ANPCYT-UUMM. (PICTO UMM-2019-00002), Research Department of the University of Mendoza Nª 13.
Compliance with Ethical Statement
All procedures were made following the Guide for the Care and Use of Laboratory Animals as adopted and promulgated by the National Institutes of Health and the EU (Eighth Edition, 2011) and approved by the Committee Institutional para el Cuidado y Uso de Animales de Laboratorio de la Universidad Nacional de Cuyo (CICUAL UNCuyo). (Aval 82/2016), Mendoza, Argentina.
Data Accessibility Statement
The authors confirm that all data underlying the findings are available without restriction. All relevant data are included in the paper.
Acknowledgements
None.
Conflict of Interest
None.
References
- Asensio JA, Cáceres A, Pelegrina LT, Sanhueza M, Scotti L, et al. (2018) Allopregnanolone alters follicular and luteal dynamics during the estrous cycle. Reprod Biol Endocrinol 16(1): 35.
- Berenguer P, Soulage C, Perrin D, Pequignot JM, Abraini JH (2003) Behavioral and neurochemical effects induced by subchronic exposure to 40 ppm toluene in rats. Pharmacol Biochem Behav 74(4): 997-1003.
- Berger M, Gray JA, Roth BL (2009) The expanded biology of serotonin. Annu Rev Med 60: 355-366.
- Bosman LW, Rosahl TW, Brussaard AB (2002) Neonatal development of the rat visual cortex: synaptic function of GABAA receptor alpha subunits. J Physiol 545(1): 169-181.
- Brown N, Kerby J, Bonnert TP, Whiting PJ, Wafford KA (2002) Pharmacological characterization of a novel cell line expressing human alpha (4) beta (3) delta GABA (A) receptors. Br J Pharmacol 136(7): 965-974.
- Buhot MC, Martin S, Segu L (2000). Role of serotonin in memory impairment. Ann Med 32(3): 210-221.
- Carver CM, Wu X, Gangisetty O, Reddy DS (2014) Perimenstrual-like hormonal regulation of extrasynaptic δ-containing GABAA receptors mediating tonic inhibition and neurosteroid sensitivity. J Neurosci 34(43): 14181-14197.
- Carver CM, Reddy DS (2013) Neurosteroid interactions with synaptic and extrasynaptic GABA (A) receptors: regulation of subunit plasticity, phasic and tonic inhibition, and neuronal network excitability. Psychopharmacology 230(2): 151-188.
- Casas S, García S, Cabrera R, Nanfaro F, Escudero C, et al. (2011) Progesterone prevents depression-like behavior in a model of Parkinson's disease induced by 6- hydroxydopamine in male rats. Pharmacol Biochem Behav 99(4): 614-618.
- Cáceres A, Vega Orozco AS, Cabrera RJ, Laconi MR (2020) "Rapid actions of the neurosteroid allopregnanolone on ovarian and hypothalamic steroidogenesis: Central and peripheral modulation." Journal of Neuroendocrinology 32(3): e12836.
- Chi JD, Odontiadis J, Franklin M (1999) Simultaneous determination of catecholamines in rat brain tissue by high-performance liquid chromatography. J Chromatogr B Biomed Sci Appl 731(2): 361-367.
- In our study, 'Olfactory bulb monoamine concentrations vary with time of day, 'published in Neuroscience, pp.247: 234-241. https://doi.org/10.1016/j.neuroscience.2013.05.040, we present novel findings that shed light on the temporal variations in olfactory bulb monoamine concentrations.
- Escudero C, Casas S, Giuliani F, Bazzocchini V, García S, et al. (2012) Allopregnanolone prevents memory impairment, effect on mRNA expression and enzymatic activity of hippocampal 3-α hydroxysteroid oxide-reductase. Brain Res Bull 87(2-3): 280-285.
- Escudero C, Giuliani F, Bernedo MM, Yunes R, Cabrera R (2019) Allopregnanolone reversion of estrogen and progesterone memory impairment: interplay with serotonin release. J Neural Transm (Vienna) 126(10): 1321-1328.
- Haller J, van de Schraaf J, Kruk MR (2001) Deviant forms of aggression in glucocorticoid hyporeactive rats: a model for 'pathological' aggression? J Neuroendocrinol 13(1): 102-107.
- Fish EW, Faccidomo S, DeBold JF, Miczek KA (2001) Alcohol, allopregnanolone, and aggression in mice. Psychopharmacology, 153(4): 473-483.
- Harvey JD, Heinbockel T (2018) Neuromodulation of Synaptic Transmission in the Main Olfactory Bulb. Int J Environ Res Public Health 15(10): 2194.
- Hernández Vázquez F, Garduño J, Hernández López S (2018) GABAergic modulation of serotonergic neurons in the dorsal raphe nucleus. Rev Neurosci 30(3): 289-303.
- Jéquier E, Lovenberg W, Sjoerdsma A (1967) Tryptophan hydroxylase inhibition: the mechanism by which p-chlorophenylalanine depletes rat brain serotonin. Mol Pharmacol 3(3): 274-278.
- Kaura V, Ingram CD, Gartside SE, Young AH, Judge SJ (2007) The progesterone metabolite allopregnanolone potentiates GABA (A) receptor-mediated inhibition of 5-HT neuronal activity. Eur Neuropsychopharmacol 17(2): 108-115.
- Khan IA, Thomas P (2004) Aroclor 1254 inhibits tryptophan hydroxylase activity in rat brains. Arch Toxicol 78(6): 316-320.
- Koolhaas JM, Coppens CM, de Boer SF, Buwalda B, Meerlo P, et al. (2013) The resident-intruder paradigm: a standardized test for aggression, violence, and social stress. J Vis Exp 4(77): e4367.
- Lima FB, Spinelli de Oliveira E (2014) How do low testosterone levels on the anatomical and behavioral repertoire of long-term enriched housing of male mice? Behav Processes, 108: 57-64.
- Locci A, Pinna G (2017) Neurosteroid biosynthesis down-regulation and changes in GABAA receptor subunit composition: a biomarker axis in stress-induced cognitive and emotional impairment. Br J Pharmacol 174(19): 3226-3241.
- Malek ZS, Dardente H, Pevet P, Raison S (2005) Tissue-specific expression of tryptophan hydroxylase mRNAs in the rat midbrain: anatomical evidence and daily profiles. Eur J Neurosci 22(4): 895-901.
- Matsumoto K, Puia G, Dong E, Pinna G (2007) GABA (A) receptor neurotransmission dysfunction in a mouse model of social isolation-induced stress: possible insights into a non-serotonergic mechanism of action of SSRIs in mood and anxiety disorders. Stress 10(1): 3-12.
- McDevitt RA, Neumaier JF (2011) Regulation of dorsal raphe nucleus function by serotonin autoreceptors: a behavioral perspective. J Chem Neuroanat 41(4): 234-246.
- Mehta AK, Ticku MK (1999) An update on GABAA receptors. Brain Res Brain Res Rev 29(2-3): 196-217.
- Meera P, Wallner M, Otis TS (2011) Molecular basis for the high THIP/gaboxadol sensitivity of extrasynaptic GABA (A) receptors. J Neurophysiol 106(4): 2057-2064.
- Miczek KA, de Boer SF, Haller J (2013) Excessive aggression as a model of violence: a critical evaluation of current preclinical methods. Psychopharmacology 226(3): 445-458.
- Miczek KA, Fish EW, De Bold JF, De Almeida RM (2002) Social and neural determinants of aggressive behavior: pharmacotherapeutic targets at serotonin, dopamine, and gamma-aminobutyric acid systems. Psychopharmacology 163(3-4): 434-458.
- Miguez J, Martin F, Aldegunde M (1991) Differential effects of pinealectomy on amygdala and hippocampus serotonin metabolism. J Pineal Res 10(2): 100-103.
- Muzerelle A, Scotto Lomassese S, Bernard JF, Soiza Reilly M, Gaspar P (2016) Conditional anterograde tracing reveals distinct targeting of individual serotonin cell groups (B5-B9) to the forebrain and brainstem. Brain Struct Funct 221(1): 535-561.
- Panzanelli P, Perazzini AZ, Fritschy JM, Sassoè Pognetto M (2005) Heterogeneity of γ-aminobutyric acid type A receptors in mitral and tufted rat central olfactory bulb. J Comp Neurol 484(1): 121-131.
- Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29(9): e45.
- Pelosi B, Pratelli M, Migliarini S, Pacini G, Pasqualetti M (2015) Generation of a Tph2 Conditional Knockout Mouse Line for Time- and Tissue-Specific Depletion of Brain Serotonin. PloS One 10(8): e0136422.
- Popova NK, Gilinsky MA, Amstislavskaya TG, Morosova EA, Seif I, et al. (2001) Regional serotonin metabolism in the brain of transgenic mice lacking monoamine oxidase A. J Neurosci Res 66(3): 423-427.
- Reddy DS (2018) GABA-Receptors Mediate Tonic Inhibition and Neurosteroid Sensitivity in the Brain. Vitam Horm pp. 107: 177-191.
- Robichaud M, Debonnel G (2004) Modulation of the firing activity of female dorsal raphe nucleus serotonergic neurons. The J Endocrinol 182(1): 11-21.
- Scott JW, Wellis DP, Riggott MJ, Buonviso N (1993) Functional organization of the main olfactory bulb. Microsc Res Tech 24(2): 142-156.
- Steinfeld R, Herb JT, Sprengel R, Schaefer AT, Fukunaga I (2015) Divergent innervation of the olfactory bulb by distinct raphe nuclei. J Comp Neurol 523(5): 805-813.
- Spigelman I, Li Z, Liang J, Cagetti E, Samzadeh S, et al. (2003) Reduced inhibition and sensitivity to neurosteroids in the hippocampus of mice lacking the GABA (A) receptor delta subunit. J Neurophysiol 90(2): 903-910.
- Stell BM, Brickley SG, Tang CY, Farrant M, Mody I (2003) Neuroactive steroids reduce neuronal excitability by selectively enhancing tonic inhibition mediated by delta subunit-containing GABAA receptors. Proc Natl Acad Sci U S A 100(24): 14439-14444.
- Strüder HK, Weicker H (2001) Physiology and pathophysiology of the serotonergic system and its implications on mental and physical performance. Part II. Int J Sports Med 22(7): 482-497.
- Takahashi A, Quadros IM, de Almeida RM, Miczek KA (2012) Behavioral and pharmacogenetics of aggressive behavior. Curr Top Behav Neurosci 12: 73-138.
- Takahashi A, Quadros IM, de Almeida RM, Miczek KA (2011) Brain serotonin receptors and transporters: initiation vs. termination of escalated aggression. Psychopharmacology, 213(2-3): 183-212.
- Takahashi A and Miczek KA (2014) Neurogenetics of Aggressive Behavior - Studies in Rodents. Curr Top Behav Neurosci 17: 3-44.
- Urban NN, Arevian AC (2009) Computing with dendrodendritic synapses in the olfactory bulb. Ann N Y Acad Sci 1170: 264-269.
- Walther DJ, Bader M (2003) A unique central tryptophan hydroxylase isoform. Biochem Pharmacol 66(9): 1673-1680.
- Wu C, Sun D (2015) GABA receptors in brain development, function, and injury. Metab Brain Dis 30(2): 367-379.
- Wu X, Gangisetty O, Carver CM, Reddy DS (2013) Estrous cycle regulation of extrasynaptic δ-containing GABA (A) receptor-mediated tonic inhibition and limbic epileptogenesis. J Pharmacol Exp Ther 346(1): 146-160.
- Yu H, Chen ZJ, Zhao JW, Duan SR, Zhao JK (2019) Olfactory Impairment and Hippocampal Volume in a Chinese MCI Clinical Sample. Alzheimer Dis Assoc Disord 33(2): 124-128.
- Zill P, Büttner A, Eisenmenger W, Möller HJ, Ackenheil M, et al. (2007) Analysis of tryptophan hydroxylase I and II mRNA expression in the human brain: a post-mortem study. J Psychiatr Res 41(1-2): 168-173.





 We use cookies to ensure you get the best experience on our website.
We use cookies to ensure you get the best experience on our website.